Hello, Goodbye: Europe loses ground as China becomes powerhouse of innovation
China has rapidly become a leading hub for innovative new medicines, overtaking Europe and putting the continent at risk of losing its relevance in biopharmaceutical innovation. However, if Europe acts swiftly, it could capitalise on recent budget cuts at key US institutions
Side A: key calls
- China will continue its rise as a powerhouse innovator.
- The US remains the most important market for biopharmaceutical innovation, but regulatory changes could threaten that in the medium term.
- Europe could capitalise on US changes, but needs to be more proactive or it will lose relevance.
You say you want a revolution: the next Pfizer will be Chinese
The ascent of China as a hub for biopharmaceutical innovation has been nothing short of spectacular. Through manufacturing specialisation, targeted government support, top science programmes and recent regulatory changes, the country has become a major hub for innovation and even surpasses the US in some areas, as evidenced by the percentage of clinical trials that now start in the country.
While this tells us nothing about the quality of clinical trials started in China, which is often debated, R&D expenditure data from the EFPIA shows that Chinese investments in fundamental research have been substantial. From 2010 to 2022, Chinese pharmaceutical R&D spending grew by 20.7% per year on average. Whereas, US spending grew by 5.5% per year and EU spending grew by 4.4% in the same period. We do not expect a similar growth rate from Chinese R&D spending until 2030, but forecast an 8% compound annual growth rate (CAGR) in this time period, which is significantly higher than our forecasts for American (5.0%) and European (4.0%) spending.
We therefore believe that the next Pfizer will be Chinese, especially if the country enacts a few more regulatory changes. This development has come at the expense of Europe, which suffers from its relative lack of scale, shallow capital markets and slower innovation. However, recent budget cuts at the National Institutes of Health (NIH) and Food and Drug Administration (FDA) could threaten the leading position of the US in the medium term (roughly 10 years). This has the potential to further accelerate the rise of China, but is also a golden opportunity for Europe and its policymakers to regain importance for pharmaceutical innovation.
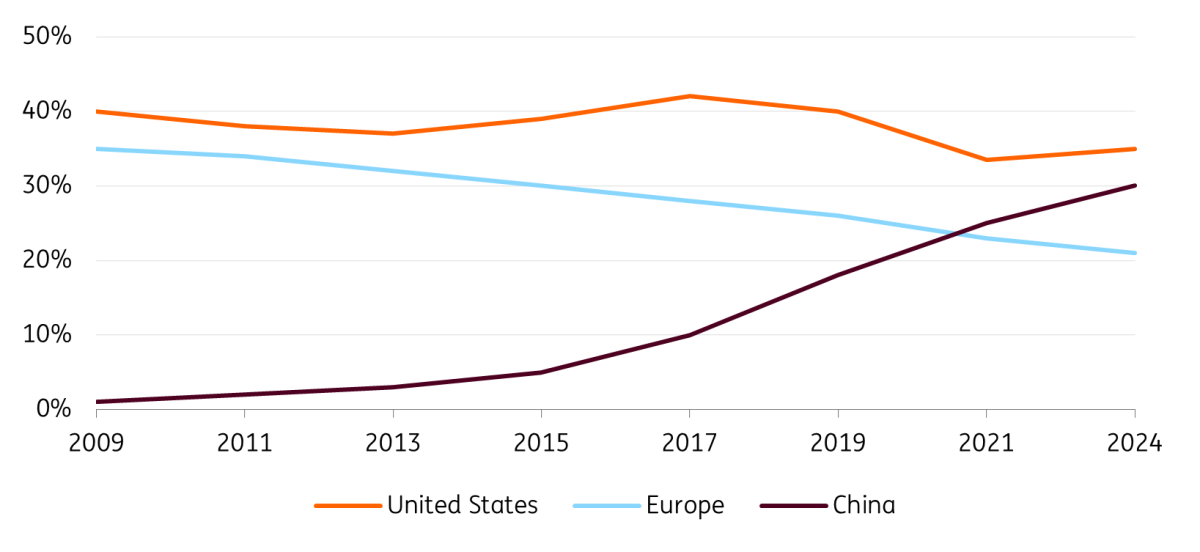
China has surpassed Europe with the percentage of started clinical trials
Clinical trial started by company location in percentages of global total
Here Comes The Sun: China already rivals the US in global drug approvals
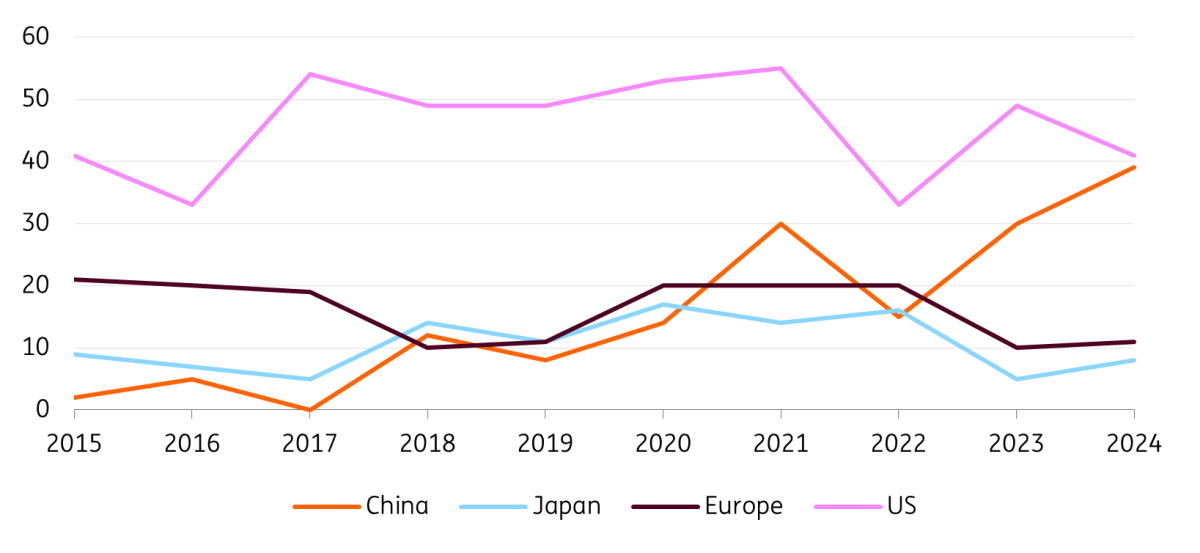
Naturally, numerous clinical trials and high R&D spending do not automatically translate into the development of new medicines. However, in recent years, China has surpassed Europe in the number of new global drug approvals, and it is edging closer to the US. This increase in new drug approvals is driven by advancements in both chemicals and biologics.
As has happened in other sectors (such as electric vehicles), China initially developed as a manufacturing hub and then gradually into a hub for innovation as well. In the late 1990s, China started focusing on producing generic drugs, and subsequently the country moved up the value chain: from supplying active pharmaceutical ingredients (APIs) to becoming a key location for outsourced biotech manufacturing to now drug development itself. At every stage, Chinese contract development manufacturing organisations (CDMOs) and clinical research organisations (CROs) gained critical expertise, resulting in the impressive number of new drugs coming out of China currently.
China has surpassed Europe in global drug approvals
Distribution of first global approvals of innovative drugs, 2015-24
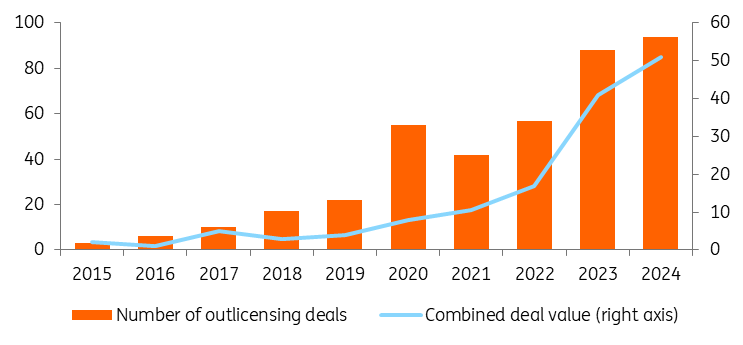
Another indicator of China’s success has been increased outlicensing deals between major pharma companies and Chinese counterparts. Outlicensing means that a company that owns a drug asset grants another company the rights to develop, manufacture and/or commercialise that drug. On average, one-third of these deals are with big pharma and above US$1bn in deal value.
The Trump Administration sees outlicensing to China as a national security threat and included restrictions for Chinese companies in the National Defence Authorisation Act at the end of last year. In spite of these restrictions, we expect outlicensing and deal value to continue to increase in 2026 and beyond.
Chinese biotech shows no signs of slowing down, and dealmaking is robust, with companies such as Merck, Bristol Myers Squibb, GSK and AstraZeneca announcing major deals recently.
Outlicensing to China has risen steeply in recent years
Number of outlicensing deals by drugmakers with Chinese counterparts and total deal value in US$bn (right axis)
Nothing’s gonna change my world: the US remains the most important innovation hub…for now
The US is by far the world’s most important pharmaceutical market: brand-name drugs are generally approved fastest in America, and they launch years earlier than in Europe, but they also cost roughly three times as much as the OECD average (while the US accounts for roughly a third of OECD GDP). As a result, the US accounts for roughly 70% of pharmaceutical profits globally. The swift and overwhelming response of pharmaceutical companies to the tariff threat underscores this importance. The announced investments do not just include investment in manufacturing but also R&D spending and M&A, which is good for the innovative capacity of the US, which mainly comes at the expense of Europe.
Furthermore, the US biotech industry is still best in class and produces high-quality new molecules at significant volumes. Before recent regulatory changes, we expected low double-digit growth until 2030. However, the Trump administration has recently enacted budget cuts at the FDA and NIH, which may hamper US innovation and growth.
First, the cuts at the FDA may threaten time to market in the US, as well as drug safety (in therapeutic areas such as psychiatric drugs), but most importantly, they threaten the predictability of the FDA, thereby adding volatility for drugmakers. This, in turn, is bad for investment in a risky sector such as biotech and could hamper innovation in the medium run. Second, the cuts at the NIH affect the ability of scientists to do fundamental research into areas that the industry deems too risky.
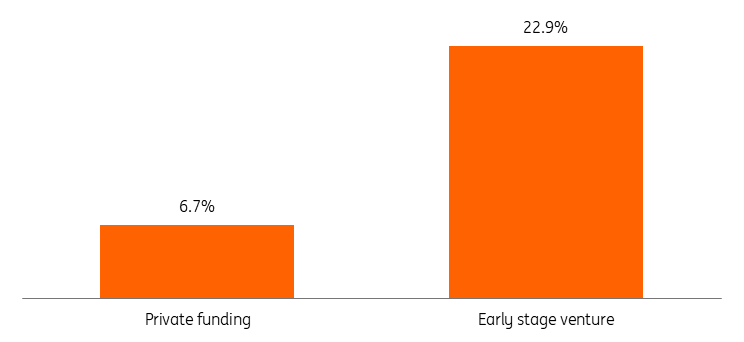
These cuts are significant: by mid-2025, 2,000 NIH research grants had been terminated (approximately $3.8bn in funding). This disrupted 383 clinical trials and 74,000 patients. These cancelled NIH grants are roughly 6.5% of the $57bn in total private funding last year (LPBI), which is not enormous. However, NIH grants are generally for commercially non-viable research projects which have shown to deliver very uneven returns (a lot fail but a select few produce blockbuster drugs). This means the distribution is skewed. In addition, much of NIH research is very early stage and total venture investment in seed through growth stages was around 16.5 billion dollars last year (Crunchbase). This means that cancelled NIH grants are roughly 23% of venture through growth funding last year, which is much more significant.
Moreover, the Trump Administration has cut research funding for many universities more broadly. These reforms will significantly hamper the innovative capacity of US biotech in the medium term. The cuts at the CDC are not material for innovation but underline the different stance of this administration towards science and public health.
The Trump Administration has also started levying $100,000 fees on H-1B visas, which hampers the ability of the US to attract and maintain top foreign scientists on which its industry heavily relies. Our outlook for US biotech has therefore declined, and we would not be surprised if US biotech growth rates drop from 2027 onwards: we forecast a 6% CAGR until 2030, which is significantly lower than before these reforms.
Cancelled NIH grants are important as a percentage of early stage venture funding
Cancelled NIH grants as a percentage of total private funding and as a percentage of early stage venture investment
However, advancements in AI may help offset some of these concerns. US firms are leading in AI development and deployment, and the Trump Administration is keen to remove obstacles that hamper swift AI deployment.
AI has two major upsides for biopharmaceutical R&D: first, AI has the potential to help discover new proteins, which could lead to the discovery of new medicines; second, AI could speed up discovery processes. So, if the potential of AI in biotech is harnessed, it could offset some of the effects of the regulatory reforms discussed above.
Yesterday: can Europe turn it around?
Europe used to be the world's biggest biopharmaceutical innovation hub: in 1990, Europe accounted for roughly half of all global pharmaceutical R&D spending compared to a third for the US. Currently, US firms account for 55% of global R&D spending, while Europe has dropped to 26% (ITIF). While European innovation spending declined, American and Asian R&D spending accelerated.
Furthermore, the percentage of clinical trials started in Europe nearly halved from roughly 35% in 2009 to 20% in 2024, and Europe’s percentage of global new drug approvals also halved: in 2024, the continent accounted for only 10% of new global drug approvals, down from 20% in 2015.
Europe is still an important centre of innovation and an important market. However, if these trends continue, it risks becoming obsolete.
European R&D spending has declined rapidly compared to the US
European and American biopharmaceutical R&D spending as a percentage of global spending in 1990 and 2025

Europe’s declining importance is the result of several European weaknesses: the continent lags China and the US in scale, speed and translational efficiency (i.e. how effectively a cell turns an mRNA molecule into a protein). Furthermore, Europe suffers from fragmented regulatory practices, differing evidence and health technology assessment (HTA) requirements, a lack of common procurement and crucially, fiscal pressure on healthcare systems due to ageing populations.
This cost pressure prevents higher prices for medicines and encourages clawback taxes which are commonplace in Europe. These subdued prices and relatively high taxes take away incentives for R&D spending in Europe. As mentioned, the US currently accounts for 70% of pharmaceutical profits and the country has a uniform set of rules, which makes it more appealing to invest there.
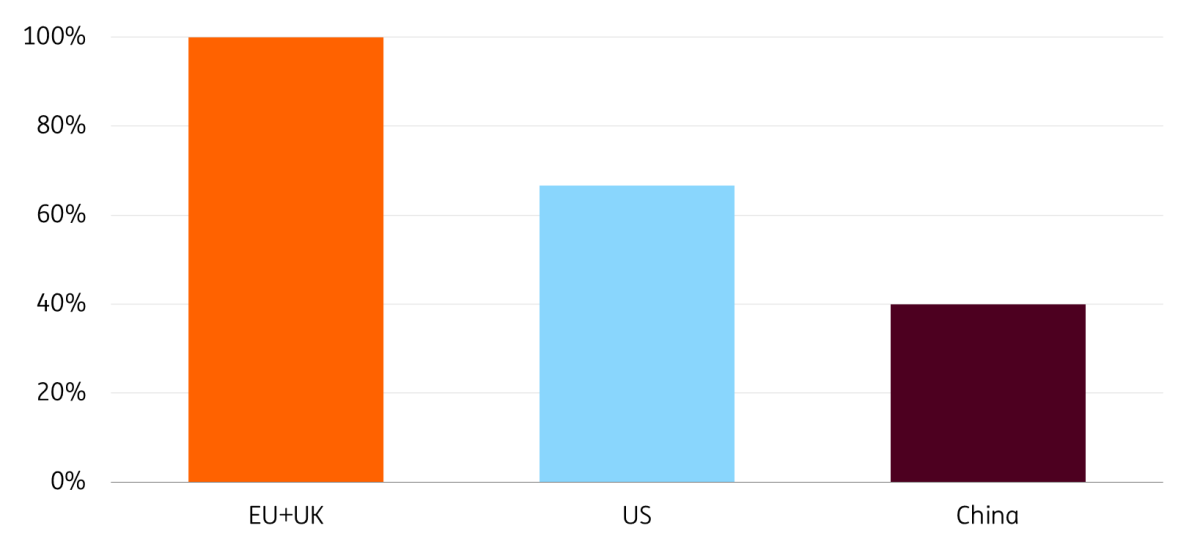
Return on investment is key, as biopharmaceutical innovation is a very costly and risky investment: on average, it takes $2bn and 10 years to take a medicine to market. Yet, Europe has all the fundamentals to become a leader in pharmaceutical innovation once again. Its researchers are cited 2.5 and 1.5 times more in top journals than their American and Chinese counterparts.
Europe’s scientists are top notch
Number of citations in top pharma journals as a percentage of ‘leader’
Europe could use the recent regulatory changes in the US to its advantage. By offering a dependable alternative to the FDA, investing in fundamental research for projects cancelled in the US and accelerating visa programmes to attract scientists, it can drastically improve its innovation capacity. In addition, the Draghi report, and national ‘follow-ups’ such as the Wennink report in the Netherlands, outline biotech as a key opportunity for high-value-added European economic growth, so it is higher on policy agendas than in previous years.
Still, to make this comeback story come to fruition, European leaders should enact policies in three critical areas:
- Consider increasing prices and decreasing clawback taxes, especially for drugs in areas of value and priority from a public health point of view. De-risking the investment case for the manufacturing of critical medication through tax breaks or subsidies should also be considered.
- Harmonised regulation on clinical trials and HTA, and expanding on the recently adopted Pharmaceutical Package. Offering time to market on par with the FDA is key, especially given FDA budget cuts.
- This is not specific to pharma, but deeper capital markets would enable more early-stage funding, which could spur innovation. In this regard, completing the Banking, Capital Markets, and Saving and Investments Union could be considered.
If these regulatory changes take effect, Europe’s pharmaceutical industry could leverage US reforms to its advantage and avoid becoming yesterday’s news.
Side B: remaining questions
The biggest question hanging over the biopharmaceutical market from an innovation perspective is to what extent Trump’s funding cuts will start to bite and how other blocs can capitalise by attracting scientists, instituting research grants and providing regulatory certainty.
If AI and private funding can fill the gap, then the effect will be limited. If other blocs attract top scientists and fund fundamental research paired with attractive opportunities for commercialisation, then the US could gradually lose its leading position.
Ultimately, the global balance of scientific leadership will hinge on whether the current uncertainty solidifies into a long-term structural shift or proves to be a temporary disruption.
This publication has been prepared by ING solely for information purposes irrespective of a particular user's means, financial situation or investment objectives. The information does not constitute investment recommendation, and nor is it investment, legal or tax advice or an offer or solicitation to purchase or sell any financial instrument. Read more
Download
Download article
20 January 2026
Pharma’s 2026 hit list: The White Album This bundle contains 6 Articles